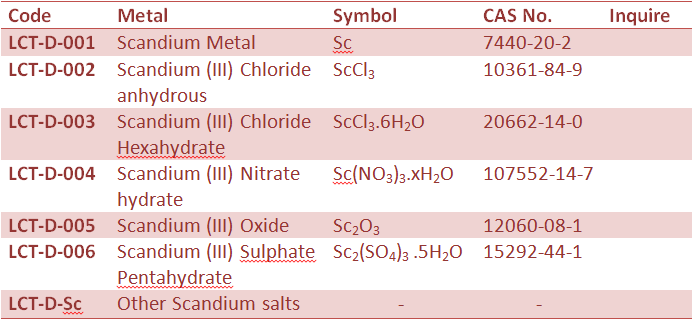
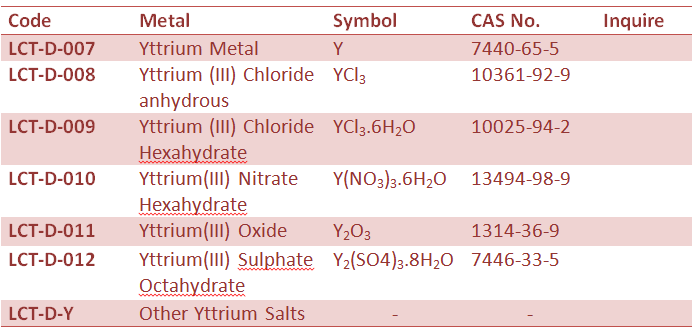
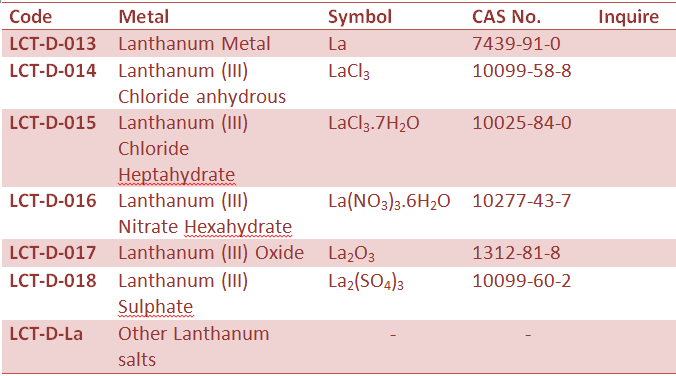
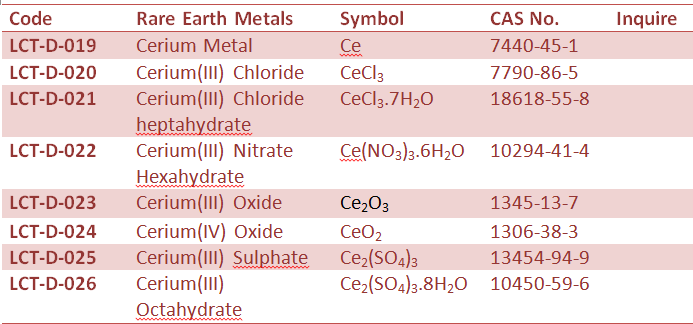
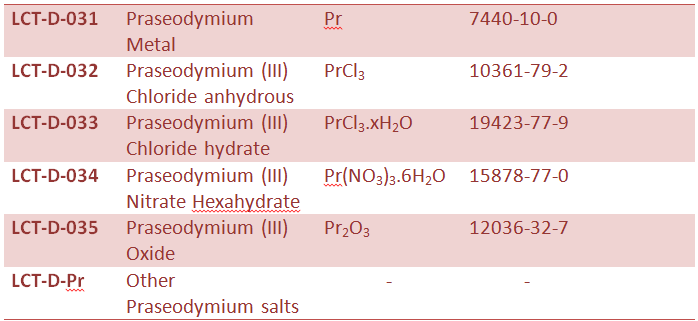
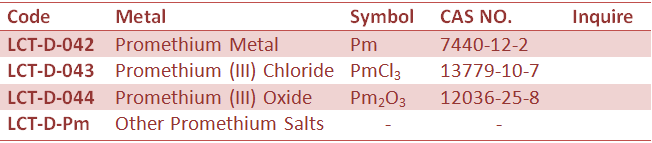
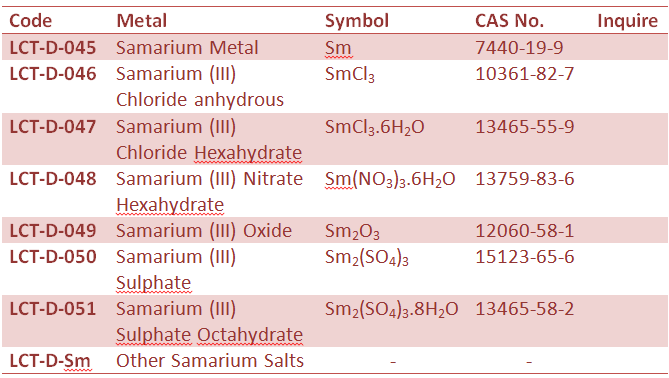
Rare Earth Metal
The rare earth elements (REE) are a group of 17 chemical (metallic) elements which appear in the periodic table. The group consists of the 15 lanthanide elements along with Yttrium and Scandium. They share many similar properties, which is why they occur together in geological deposits. The 17 REEs are found in all REE deposits but their distribution and concentrations vary. They are referred to as ‘rare’ because it is not common to find them in commercially viable concentrations. REEs generally fall into one of two categories – light rare earths (LREE) and heavy rare earths (HREE), with varying levels of uses and demand. REE mineral deposits are usually rich in either LREE or HREE, but rarely contain both in significant quantities. In general, they are vital to some of the world’s fastest growing markets: clean energy and high technology.
Silvery-white or gray in colour, these metals have a high lustre and tarnish readily when exposed to air. REEs are found in most everyday applications because of their unique chemical and physical properties.
New applications have arisen consistently over the past 50 years CIBC, including important environmental innovations such as catalytic converters and the development of permanent magnets which have enabled greater efficiency, miniaturization, durability and speed in electric and electronic components. Substitutes exist, but rarely work as effectively.
Silvery-white or gray in colour, these metals have a high lustre and tarnish readily when exposed to air. REEs are found in most everyday applications because of their unique chemical and physical properties.
New applications have arisen consistently over the past 50 years CIBC, including important environmental innovations such as catalytic converters and the development of permanent magnets which have enabled greater efficiency, miniaturization, durability and speed in electric and electronic components. Substitutes exist, but rarely work as effectively.